In-Situ Gas Analysis of Lithium-Ion Batteries
There are numerous side reactions occurring within a battery over its lifetime that adversely affect its performance, whether during operation or storage. As many of these side reactions are associated with the production of gaseous species, in-situ techniques that allow for the identification and quantification of such species can be powerful. To this end, our group provides setups that allow for in-situ gas analysis which utilize mass spectrometry, infrared spectroscopy, and pressure measurements on custom electrochemical cells.
Differential Electrochemical Mass Spectrometry/Differential Electrochemical Infrared Spectroscopy (DEMS/DEIRS)
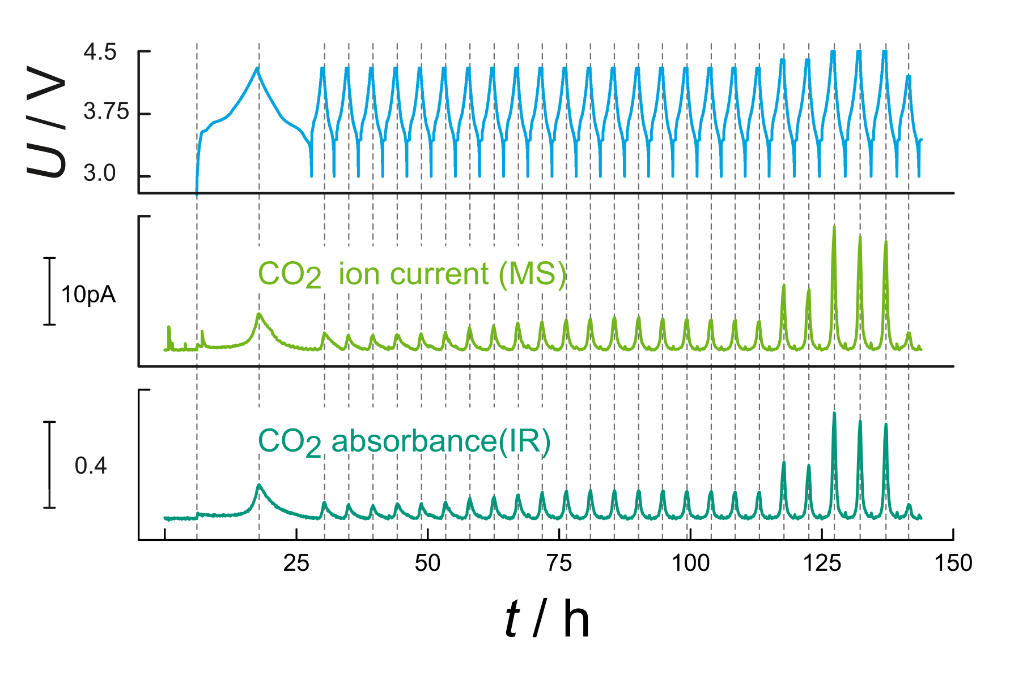
Operation principle: A mass flow controller (MFC) provides an inert carrier gas that flushes the cell during operation. Gases that evolve are directly carried out and analyzed via mass spectrometry (MS) and infrared spectroscopy (IR). A second MFC provides a calibration gas with known gas composition to quantify the detected gas amounts. A photograph of the setup is depicted in Figure 1, while a typical measurement for an NCM523 cell is depicted in Figure 2. For clarification, only the CO2 signals are shown. The measurement shows the strong correlation between electrochemical cycling and gas evolution.
In-Situ Pressure Analysis
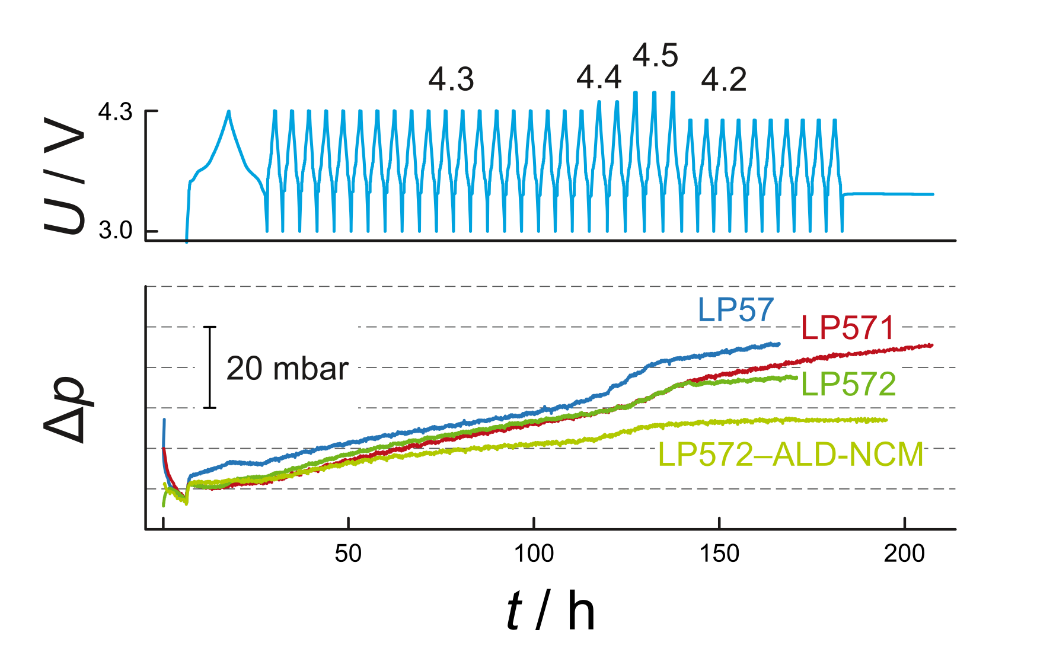
Operation principle: A custom-made cell is placed inside a thermostat, allowing for battery operation between 0 °C and 60 °C. Additionally a pressure sensor is attached to the cell, recording pressure evolution during cycling. A typical measurement is depicted in Figure 3.
Fields of application for in-situ gas/pressure analysis:
- Electrolyte additives
- Temperature-dependent aging
- Active material coatings
- Next-generation materials
- Long-term cycling studies