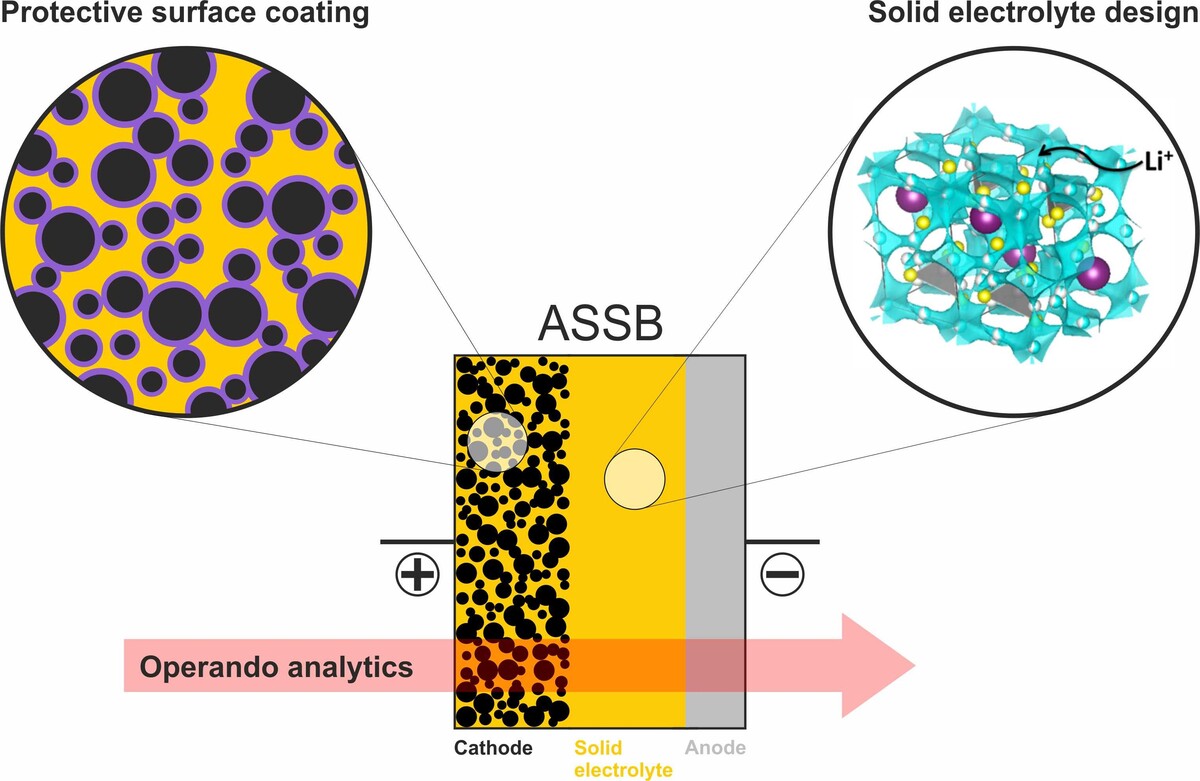
All-Solid-State Batteries
The ever-increasing demand for high-energy-density energy storage devices, which enable electric powered transportation, has triggered the development of all-solid-state batteries (ASSBs). In principle, replacing the liquid electrolyte of state-of-the-art lithium-ion batteries should allow for higher energy and power densities. However, their commercial implementation is hindered by several obstacles, mainly related to the electrochemical instability of the solid electrolyte towards the electrodes. Our research is focused on understanding detrimental side reactions in ASSBs from a fundamental point of view and tackle these issues through rational surface modifications as well as solid electrolyte design/improvement.
Advanced Analytics
We aim at studying and characterizing processes occurring in ASSBs at cell level as well as on an atomistic scale at the solid electrolyte/electrode interfaces. This involves an understanding of structural degradation, influence of cell construction/composition, and development of setups allowing for operando measurements.
Protective Surface Coatings
In order to prevent severe side reactions between the cathode material and the solid electrolyte, protective coating layers are required. Following this line, optimization and development of new coating chemistries, along with a thorough understanding of composition-property relationships, are pursued. This is done using a wide range of synthetic techniques such as sol-gel and atomic layer deposition, complemented by various characterization tools, including electron microscopy, X-ray absorption, and infrared spectroscopy.
Solid Electrolyte Design
Because the stability of the solid electrolyte is a serious problem in ASSBs, our objective is to gain insight into the degradation mechanisms occurring at the electrode/electrolyte interfaces and tailor the electrochemical stability/ionic conductivity of the solid electrolyte.