Thioacetate‐Based Initiators for the Synthesis of Thiol‐End‐Functionalized Poly(2‐oxazoline)s
-
Author:
G. G. Alvaradejo, M. Glassner, R. Kumar, V. Trouillet, A. Welle, Y. Wang, V. R. de la Rosa, S. Sekula‐Neuner, M. Hirtz, R. Hoogenboom, G. Delaittre
-
Source:
Macromol. Rapid Commun. 41 (2020) 2000320
- Date: 16.08.2020
-
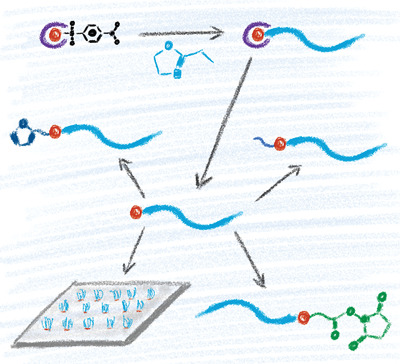
New functional initiators for the cationic ring‐opening polymerization of 2‐alkyl‐2‐oxazolines are described to introduce a thiol moiety at the α terminus. Both tosylate and nosylate initiators carrying a thioacetate group are obtained in multigram scale, from commercial reagents in two steps, including a phototriggered thiol–ene radical addition. The nosylate derivative gives access to a satisfying control over the cationic ring‐opening polymerization of 2‐ethyl‐2‐oxazoline, with dispersity values lower than 1.1 during the entire course of the polymerization, until full conversion. Cleavage of the thioacetate end group is rapidly achieved using triazabicyclodecene, thereby leading to a mercapto terminus. The latter gives access to a new subgeneration of α‐functional poly(2‐oxazoline)s (butyl ester, N‐hydroxysuccinimidyl ester, furan) by Michael addition with commercial (meth)acrylates. The amenability of the mercapto‐poly(2‐ethyl‐2‐oxazoline) for covalent surface patterning onto acrylated surfaces is demonstrated in a microchannel cantilever spotting (µCS) experiment, characterized by X‐ray photoelectron spectroscopy (XPS) and time‐of‐flight secondary‐ion mass spectrometry (ToF‐SIMS).
