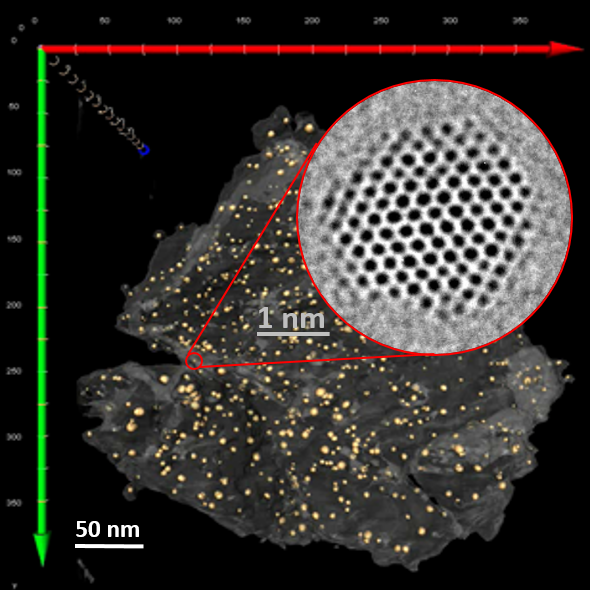
Continuous flow synthesis of atom-precise platinum clusters
A modular continuous-flow synthesis strategy was developed to produce atomically precise platinum nanoclusters with controlled size and composition. The approach employs a cascade of continuous stirred-tank reactors that enables stable reaction conditions, precise control over nucleation and growth, and improved reproducibility compared with conventional batch synthesis methods. This reactor configuration allows the sequential formation and stabilization of platinum clusters under steady-state conditions, facilitating scalable production of well-defined nanoscale structures relevant for catalytic and nanomaterial applications.
Comprehensive structural characterization confirmed the formation of highly uniform clusters with well-defined atomic structure. Advanced electron microscopy techniques provided direct visualization of the nanoclusters, enabling the analysis of particle morphology, size distribution, and crystallinity at the nanoscale. High-resolution imaging revealed structurally homogeneous clusters and allowed the identification of lattice features consistent with metallic platinum. These observations, supported by complementary analytical techniques such as mass spectrometry, verified the atom-precise nature of the synthesized clusters and demonstrated the effectiveness of the continuous-flow approach for generating structurally controlled metallic nanoclusters.
Details and further work are published at:
- Schmitt, C., Da Roit, N., Neumaier, M., Maliakkal, C. B., Wang, D., Henrich, T., Kübel, C., Kappes, M., & Behrens, S. (2024). Continuous flow synthesis of atom-precise platinum clusters. Nanoscale Advances, 6, 2459–2468. https://doi.org/10.1039/D4NA00074A
Disclosing the leaching behaviour of Pd@CMK3 catalysts in formic acid decomposition by electron tomography
Understanding the leaching behaviour of Pd@CMK3 catalysts during formic acid decomposition requires examining how pore confinement, nanoparticle mobility, and reactor hydrodynamics co-govern catalyst stability and performance. The article demonstrates that leaching is not a singular degradation mechanism but arises from a dynamic balance between Pd dissolution, particle sintering, and redeposition processes that unfold differently depending on the reaction environment and the accessibility of mesoporous channels within the CMK3 support. In batch reactors, where reactants and catalyst coexist in a closed volume, dissolved Pd species encounter restricted diffusion paths and prolonged residence times, conditions that encourage redeposition onto the internal and external surfaces of the mesoporous carbon. This enhances the likelihood of retention of active metal, even as nanoparticle size distributions evolve through ripening and aggregation, illustrating how confinement within sub-nanometer to nanometer pores can partially mitigate metal loss by physically and kinetically stabilizing ultrafine particles. By contrast, continuous-flow reactors promote pronounced leaching through persistent solvent flux, which continuously extracts dissolved Pd from the porous matrix and limits redeposition, leading to severe depletion of internal particles and preferential redeposition only near external surfaces, where fluid transport is more accessible. This interplay underscores that catalyst architecture—specifically the spatial distribution of ultrafine nanoparticles relative to internal versus external pore networks—must be engineered in concert with reactor design, as identical formulations can display markedly different degradation pathways depending on operational mode. Moreover, electron tomography is a indispensable three-dimensional analytical technique capable of resolving the nuanced redistribution of nanoparticles at nanometric scale, revealing structural evolutions that conventional 2D characterization obscures and establishing reliable correlations between nanoscale structural change and catalytic durability. Collectively, these insights emphasize that controlling metal leaching in heterogeneous catalysts is not solely a function of chemical composition, but also a consequence of physical confinement, fluid dynamics, and surface accessibility—all critical parameters for designing next-generation catalysts optimized for hydrogen release from formic acid and other liquid organic hydrogen carriers.

Details and further work are published at:
- Huang, X.; Barlocco, I.; Villa, A.; Kübel, C.; Wang, D. Disclosing the Leaching Behaviour of Pd@CMK3 Catalysts in Formic Acid Decomposition by Electron Tomography. Nanoscale Advances, 2023, 5, 1141–1151. DOI: 10.1039/d2na00664b.
Precisely Picking Nanoparticles by a “Nano-Scalpel” for 360° Electron Tomography
A fundamental challenge in modern catalysis research—particularly when dealing with ultrafine metal nanoparticles—is establishing a reliable correlation between nanoscale structure and catalytic performance, a task that becomes increasingly complex when the active phase is highly dispersed or embedded within hierarchical supports. The methodological advancement by introducing a “nano-scalpel” approach to isolate individual or clustered nanoparticles for full-range 360° electron tomography, thereby eliminating the missing wedge problem that has long hindered accurate three-dimensional reconstruction. This achievement is not merely a technical refinement; it directly strengthens catalytic science by enabling researchers to visualize ultrafine metal particles—such as Pt on TiO₂ hollow cages—in their true three-dimensional morphology, allowing for precise evaluation of particle geometry, support interactions, porosity and confinement effects, all of which play decisive roles in catalytic turnover frequency, selectivity, and stability. In heterogeneous catalysis, features such as facet exposure, particle curvature, and interfacial contact area govern adsorption energies and activation barriers; hence, accurate 3D reconstructions enable a more rigorous interpretation of site-specific reactivity.

Moreover, the capability to extract and mount selected nanoparticles without mechanical or ion-beam-induced damage is particularly consequential for ultrafine systems where particle size approaches a regime where even minor structural perturbations can dramatically alter catalytic pathways via quantum size effects or altered electronic structures. Full rotation tomography therefore becomes a catalyst-design tool: resolving mesoporous diffusion networks, hollow supports, and anisotropic nanoparticles with minimized reconstruction artifacts allows the derivation of more trustworthy structure–performance relationships, informing rational strategies in catalyst synthesis such as controlled alloying, tailored supports, or confinement engineering for reactions including hydrogen generation, selective oxidation, or CO₂ conversion. By bridging methodological rigor with catalytic relevance, this approach expands the analytical capabilities required to decipher the active nature of ultrafine catalysts and enhances the predictive power of structure-guided catalyst development.
Details and further work are published at:
- Huang, X.; Tang, Y.; Kübel, C.; Wang, D. Precisely Picking Nanoparticles by a “Nano-Scalpel” for 360° Electron Tomography. Microscopy and Microanalysis, 2022, 28, 1981–1988. doi:10.1017/S1431927622012247.
Alloyed Supported Catalysts in Formic Acid Decomposition
The catalytic improvement observed in Pd–Au alloyed systems reflects a sophisticated interplay between nanoscale structure, surface chemistry, and reaction energetics, which collectively determine how efficiently formic acid can be converted into hydrogen under mild conditions. The introduction of gold into palladium matrices does more than merely dilute active sites; it fundamentally modifies Pd’s electronic environment through electron transfer processes that alter adsorption strengths and reaction pathways. This modification is evident in the binding energy shifts and surface composition changes revealed through XPS, where Au donates electron density to Pd and facilitates the preferential exposure of palladium atoms at the nanoparticle surface. Such electronic restructuring suppresses CO poisoning by weakening the adsorption of poisoning intermediates relative to dehydrogenation species, a critical limitation in monometallic Pd catalysts. At the same time, STEM and EDX analyses confirm that alloying yields homogeneous and stable Pd–Au distributions, supporting the premise that compositional tuning at the nanoscale is central to optimizing catalytic behavior. Complementary DFT calculations illuminate these experimental observations with atomistic insight: bimetallic Pd₉Au₆ clusters not only bind formic acid exothermically but also steer decomposition along the more desirable dehydrogenation route, producing CO₂ and H₂ while energetically disfavoring the dehydration pathway. This preference emerges from a delicate balance of ligand and strain effects, which reduce ensemble arrangements prone to CO formation and stabilize reaction intermediates. Furthermore, the stronger adhesion between bimetallic nanoparticles and carbon nanofiber supports—compared to monometallic analogues—confers resistance to sintering and agglomeration, ensuring sustained catalytic performance over multiple reaction cycles without significant loss of activity or selectivity. Collectively, these findings position Pd–Au alloyed catalysts as strategically engineered systems in which nanoscale alloying, support interactions, and electronic tuning converge to elevate hydrogen production efficiency, demonstrating how rational design principles can transform molecular-scale mechanisms into tangible advancements for sustainable energy applications.

Details and further work are published at:
- Barlocco, I.; Capelli, S.; Lu, X.; Bellomi, S.; Huang, X.; Wang, D.; Prati, L.; Dimitratos, N.; Roldan, A.; Villa, A. Disclosing the Role of Gold on Palladium–Gold Alloyed Supported Catalysts in Formic Acid Decomposition. ChemCatChem, 2021, 13, 4210–4222. https://doi.org/10.1002/cctc.202100886
Highly Active Oxidation Catalysts
In advanced heterogeneous catalysis, several interrelated concepts define the performance and stability of noble-metal systems. The notion of noble-metal dispersion describes how Pd exists either as atomically dispersed species or as nanoclusters whose nuclearity dictates reactivity; ceria–metal interactions refer to the strong anchoring of Pd on CeO₂, which stabilizes oxidized species yet can also drive redispersion under oxidizing conditions; the cluster formation threshold reflects the local surface noble-metal concentration required for transition from isolated atoms to catalytically competent PdOₓ clusters; and spatial confinement on nano-islands—here realized by embedding CeO₂ domains within an Al₂O₃ matrix—limits Pd mobility, mitigating sintering and over-dispersion, while enabling in situ cluster formation even at low overall loadings (0.5 wt%).

Details and further work are published at:
- Gashnikova, D., Maurer, F., Sauter, E., Bernart, S., Jelic, J., Dolcet, P., Maliakkal, C. B., Wang, Y., Wöll, C., Studt, F., Kübel, C., Casapu, M., & Grunwaldt, J.-D. (2024). Highly Active Oxidation Catalysts through Confining Pd Clusters on CeO₂ Nano-Islands. Angewandte Chemie International Edition, 63(35), e202408511. https://doi.org/10.1002/anie.202408511
Influence of the CeO2 Morphology and Initial Pd−Pt Interaction Degree on Catalyst Activity and Stability
The catalytic behavior of Pd–Pt systems supported on ceria cannot be evaluated solely on the basis of composition; rather, it must be interpreted through the combined lens of support morphology, metal–metal interaction, and nanoscale structural evolution, all of which are most convincingly revealed through electron microscopy. Ceria nanocubes and nanorods present markedly different surface terminations and defect populations, influencing the anchoring energy landscape for Pd and Pt species and thereby dictating the formation of either alloyed clusters or more dispersed ionic species. These morphological attributes govern the density and mobility of oxygen vacancies—central actors in CO oxidation—and thus directly regulate the redox cycling capacity of the support–metal interface. Transmission electron microscopy proves vital in this context, allowing the authors to distinguish not only particle size distributions but also degree of alloying, metal-support contact geometry, and the progressive restructuring of active phases after oxidative or reductive treatments. High-resolution TEM and STEM–EDX mapping expose how nanocubes stabilize sub-nanometric Pd–Pt ensembles with intimate metal–support bonding, whereas nanorods—with inherently higher defect densities—favor more extensive metal redispersion under reaction conditions, which can paradoxically reduce the number of structurally optimal active sites for low-temperature CO conversion. Furthermore, comparative TEM analyses before and after long-term operation reveal sintering resistance in cube-supported systems and highlight the susceptibility of rod-supported catalysts to undesirable particle growth or over-dispersion, phenomena that would remain speculative without direct nanoscale imaging. These insights underscore that catalyst stability and activity must be assessed not only in terms of nominal loading and alloying degree but through an appreciation of how nanostructure responds dynamically to thermal and chemical stimuli—an understanding that is only accessible via TEM’s capability to visualize atomic-scale transformations. Successful catalyst design hinges on selecting ceria morphologies that foster a controlled Pd–Pt interaction degree at the outset and maintain it throughout operation, enabling sustained performance in oxidation environments relevant to emissions control and broader industrial catalysis.

Figure 1. HAADF-STEM images of the fresh Pd-Pt bimetallic catalysts, namely, PdPt-MF1-com (a),PdPt-MF1-rod(d),PdPt-MF1 cub(g),and PdPt-MF2-cub (j). EDX elemental mapping for Pd(b,e, h,k) and Pt (c,d, i, l) corresponding to the HAADF-STEM images.
Details and further work are published at:
- De Giacinto, A., Dolcet, P., Wang, D., Czechowsky, J., Maliakkal, C. B., Kübel, C., Behrens, S., Grunwaldt, J.-D., Gross, S., & Casapu, M. (2025) Influence of the CeO₂ Morphology and Initial Pd–Pt Interaction Degree on Catalyst Activity and Stability. Journal of Physical Chemistry C, 129, 14866–14889.